Geron Corp. (NASDAQ: GERN) is a commercial-stage biopharmaceutical company dedicated to changing the lives of patients by changing the course of blood cancers. It develops and sells targeted oncology treatments, with a focus on telomerase inhibition to treat rare blood cancers.
What Does GERN Do?
GERN focuses on developing and commercializing first-in-class therapies that target the uncontrolled proliferation of malignant cells in hematologic malignancies.
-
- Key Commercial Product: RYTELO (imetelstat) is the company’s lead asset. It is the first and only FDA-approved telomerase inhibitor for adult patients with lower-risk myelodysplastic syndromes (MDS) who have transfusion-dependent anemia.
- MDS is a type of blood cancer where the bone marrow doesn’t make enough healthy blood cells.
- Scientific Approach: GERN’s technology targets telomerase activity, which is typically increased in malignant stem and progenitor cells in the bone marrow. By inhibiting this enzyme, RYTELO aims to reduce the survival of these cancer cells and improve patient outcomes.
- Patient Impact: For patients, RYTELO means potentially fewer blood transfusions, which are time-consuming and carry risks of iron overload.
- Clinical Pipeline: GERN is advancing RYTELO for other high-need indications, including a pivotal Phase 3 trial (IMpactMF) for relapsed/refractory myelofibrosis (MF).
- Key Commercial Product: RYTELO (imetelstat) is the company’s lead asset. It is the first and only FDA-approved telomerase inhibitor for adult patients with lower-risk myelodysplastic syndromes (MDS) who have transfusion-dependent anemia.
What Value does GERN offer?
-
- First-in-Class Status: RYTELO is the only approved therapy in its class, providing a unique treatment option for MDS patients who have failed or are ineligible for erythropoiesis-stimulating agents (ESAs).
- Infusion Moat: As an intravenous (IV) infusion administered in clinics, RYTELO benefits from a “buy-and-bill” infrastructure that creates a defensive moat against emerging oral competitors.
- “buy-and-bill“: allows doctors to get reimbursed faster, thereby providing them with an incentive to prescribe it.
- Established Commercial Presence: Since its 2024 launch, RYTELO has gained significant traction, achieving $183.6 million in net revenue for 2025.
- Global Expansion Potential: With recent approvals in both the U.S. and the European Union (EU), GERN is positioned to tap into international markets starting in 2026.
What is the Market’s Take and Why?
-
- Strong Revenue Guidance: Management projects 2026 net product revenue between $220 million and $240 million, representing roughly 25% y/y growth.
- High Institutional Conviction: Approximately 74% of GERN shares are held by institutions, signaling deep professional confidence in the company’s long-term oncology strategy.
- Pathway to Profitability: 2026 is viewed as a transitional year where narrowed losses set the stage for a stronger 2027.
- With projected operating expenses of $230M–$240M and revenue potentially reaching $240M, the company is nearing an operating breakeven point.
- The late-2025 restructuring plan reduced the workforce by one-third to lower operating expenses and accelerate the path toward profitability by 2027, effectively “de-risking” the stock for long-term holders.
-
- Stagnating Sales Growth: Despite the strong annual totals, recent quarterly performance has been flat, with Q4 2025 revenues ($48 million) missing consensus estimates.
- Ongoing Net Losses: While narrowing, Geron is still expected to report a GAAP net loss for FY 2026.
- Estimates are for a full-year non-GAAP EPS loss of approximately -$0.046 to -$0.07 per share.
- GERN’s 44% debt-to-equity ratio, used to fund the RYTELO launch, remains a point of concern for conservative investors.
- Concentration Risk: RYTELO, RYTELO, RYTELO. That’s it! GERN’s valuation is heavily tied to the commercial success and pipeline expansion of this single asset.
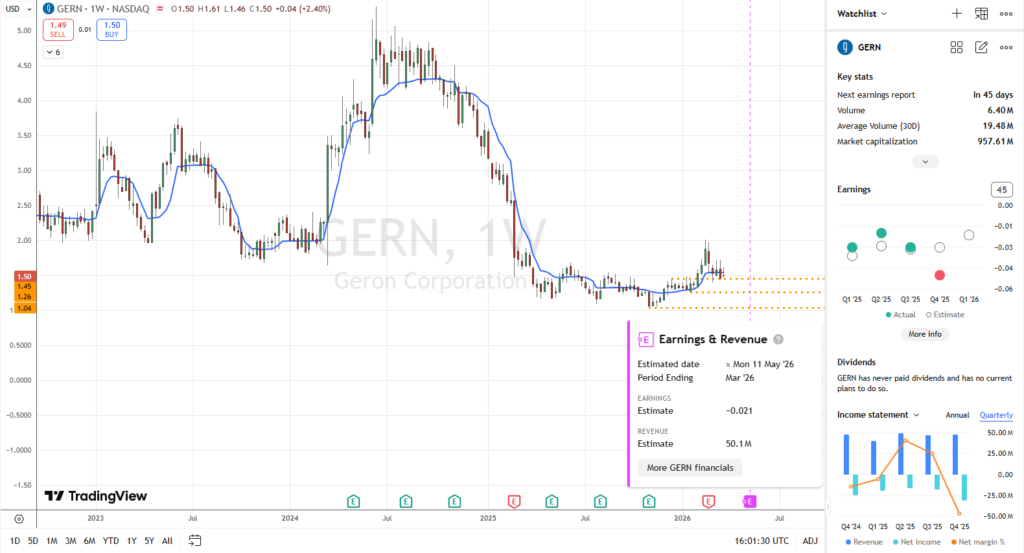
Upcoming Catalyst(s)
-
- Q1 2026 Earnings:
- Revenue Consensus: $50.11M – $50.97M. This will be the first major test of GERN’s progress toward its $220M–$240M full-year guidance and prove its commercial execution capability.
- EPS Forecast: Estimates are for a net loss of -$0.02 to -$0.03 per share.
- Focus: Investors will be looking for signs that reallocating resources toward the commercial sales team (following the restructuring) is successfully driving higher RYTELO adoption.
- European Launch Strategy (H1 2026): Updates on the commercialization strategy for RYTELO in the European Union are expected in the first half of the year.
- IMpactMF Interim Analysis (H2 2026):
- Clinical Significance: This is the first Phase 3 trial in relapsed/refractory myelofibrosis to use overall survival as its primary endpoint.
- Expectation: A positive readout could prove RYTELO’s disease-modifying potential and significantly expand its market.
- Q1 2026 Earnings:
(** NOT FINANCIAL ADVICE **)
GERN is a speculative long-term play, at best. Buying DEEP (Jan 2027) OTM CALLS might be the way to go as a bet on its ability to turn profitable.