Precigen Inc. (NASDAQ: PGEN) is a biopharmaceutical company that focuses on advancing the next generation of gene and cell therapies to treat complex, high-need diseases. The company emphasizes a “Great Science, Greater Cause” approach, aiming to create life-saving, cost-conscious therapies for patients with significant unmet medical needs.
What Does PGEN Do?
Precigen develops precision medicines using proprietary technology platforms to target intractable diseases.
-
- Key Commercial Product: PAPZIMEOS is the company’s lead asset. It is the first and only FDA-approved therapy for adults with Recurrent Respiratory Papillomatosis (RRP), a rare disease caused by HPV 6 or 11. RRP often requires patients to undergo dozens, sometimes hundreds, of surgeries over a lifetime. A non-surgical cure would be a game-changer!
- Technology Platforms:
- AdenoVerse: Utilizes a library of proprietary adenoviral vectors for efficient gene delivery.
- UltraCAR-T: A non-viral multigenic delivery system that enables a rapid manufacturing process for CAR-T therapies. Traditional CAR-T requires a patient to wait 3–4 weeks for their cells to be modified in a lab — PGEN can do it in 24 hours!
- Clinical Pipeline: The company is advancing therapies for various cancers (e.g., PRGN-3005 for ovarian cancer, PRGN-3006 for AML) and autoimmune conditions like Type 1 Diabetes (AG019).
What Value does PGEN offer?
-
- First-Mover Advantage: With the approval of PAPZIMEOS, Precigen holds an exclusive market window for the only approved treatment for adult RRP.
- Scalable Manufacturing: Its UltraCAR-T platform reduces manufacturing time from weeks to just one day, potentially lowering costs and allowing for decentralized production at medical centers.
- Commercial Momentum: Rapid adoption is evident, with patient hub enrollment doubling recently and health plan coverage reaching approximately 215 million lives in the US. (This is a measure of potential access, not actual approvals)
What is the Market’s Take and Why?
Investors have a polarized view of Precigen, reflecting its high-risk, high-reward transition from a biotech developer to a commercial entity.
Bullish
-
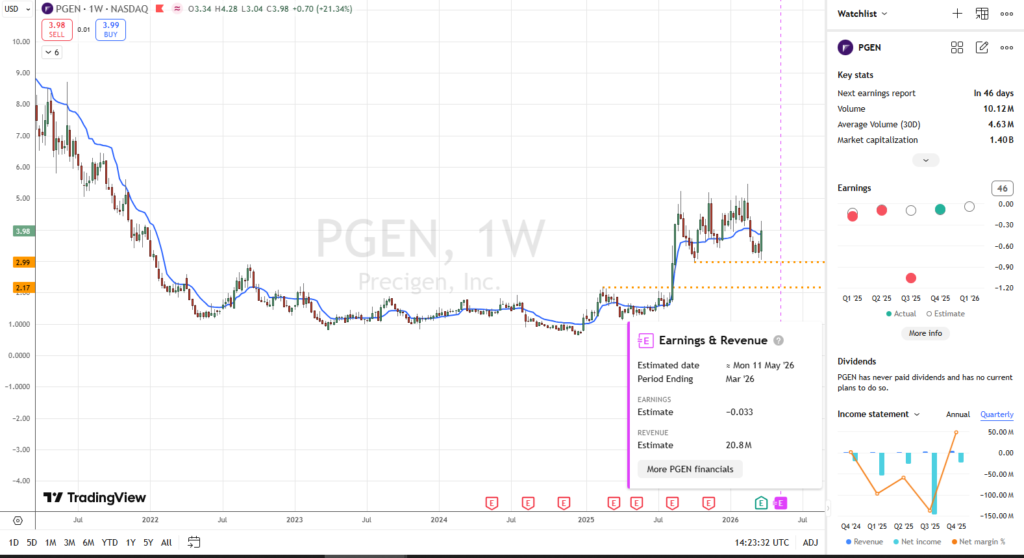
- Commercial Momentum: The market is encouraged by the rapid adoption of PAPZIMEOS. The company’s Q4 2025 earnings report outperformed expectations, and management’s guidance for Q1 2026 projects over $18 million in revenue—a massive sequential increase.
- Path to Profitability: Goal of reaching cash-flow breakeven by the end of 2026, which would significantly “de-risk” the company’s financial future.
- Market Dominance: As the only approved therapy for adult RRP, PAPZIMEOS currently enjoys a monopoly position, allowing for premium pricing and rapid market penetration.
Bearish
-
- Financial Instability: Despite rising revenue, PGEN reported a staggering $429.6 million net loss for 2025. Management attributes this to one-time $318.5 million in non-cash charges related to preferred stock warrants that will not recur. Still, there is fear of potential share dilution if cash-flow targets are missed.
- Concentration Risk: The current valuation relies almost entirely on the success of a single asset (PAPZIMEOS). Any setback in commercial roll-out or insurance reimbursement could cause the stock to plummet.
- Upcoming Competition: A direct competitor (Inovio Pharmaceuticals’ INO-3107) is currently awaiting a regulatory decision in mid-2026, which could challenge PGEN’s market share.
- Valuation Concerns: Some DCF (Discounted Cash Flow) models suggest a much lower fair value (near $1.00 or less) compared to current market prices, leading some to believe the stock is overextended.
Upcoming Catalyst(s)
-
- April 1, 2026: Implementation of a permanent J-code (J3404) for PAPZIMEOS, which is expected to streamline the claims process and accelerate patient access. J-code isn’t just a ‘billing thing’; It’s a ‘Buy-and-Bill’ trigger that allows doctors to get reimbursed faster thereby providing them incentive to prescribe it.
- Q1 2026 Revenue Reporting: Guidance for product sales is over $18 million — which would represent a near-doubling of their entire 2025 performance in just 90 days — and would speak to their commercial execution.
- European Expansion: Continued review and potential approval by the European Medicines Agency (EMA) for PAPZIMEOS.
- Pipeline Progress: Updates on Phase 1/1b studies for PRGN-3006 (AML) and PRGN-3007 (ROR1+ tumors) are expected to drive sentiment beyond the lead asset.
(** NOT FINANCIAL ADVICE **)
PGEN’s massive EPS beat gives credence to its ‘Science’. This might be enticing enough to BUY deep (Jan 2027 or 2028) OTM CALLS as a bet on its ‘Sales Execution.’